NOTES: All of the best practices described below have been developed and hands-on tested in an actual dietary intervention study as feasible and minimally disruptive in a commercial-standard kitchen.
The kitchen must be treated as a laboratory; a recipe is a protocol; foods are reagents; every item used in preparation and cooking is equipment and instrumentation; detailed notes must be captured on every item, step, and procedure.
This ad-free article is made possible by the financial support of the
Center for Research on Environmental Chemicals in Humans: a 501(c)(3) non-profit.
Please consider making a tax-deductible donation for continued biomedical research.
CARDINAL COROLLARY#1: You must know, and disclose: the exact composition and source of every ingredient, item, and piece of equipment.
CARDINAL COROLLARY #2: The exact ingredients, items, and pieces of equipment must be readily available to the broadest geographical spectrum of investigators.
CARDINAL COROLLARY #3: The preparation environment must be precisely and completely described in a manner that it can be duplicated by other investigators.
CARDINAL COROLLARY #4: Rely only on data that you have observed and collected. Do not use self-reported or memory-based information or rely on un-monitored test subject compliance.
Accuracy and replicability depend upon using precise methods under identical conditions using identical materials, reagents, apparatus, and test subjects. Unfortunately, human dietary intervention studies located in a search of the scientific literature are fatally confounded by the failure to employ basic scientific methods that are standard in bench experiments.
In addition to these shortcomings, dietary intervention studies treat the kitchen as a black box to be provided by caterers, institutional kitchens, or even test subject memories. (Black Box meals: A look at five dietary intervention studies. )
Most studies involving BPA and phthalates have given general instructions to food preparers to use only “fresh” ingredients and to avoid any plastic contact. However, specific food sources and the details of their production are not available. Neither are specific cooking protocols nor the identities of the utensils, pots, pans, cleaning regimen (surfactants an issue), recipes, or ingredients (processing & methyl contributors an issue).
Data regarding those conditions cannot account for the wide variability of contamination in the growing, processing, and sourcing stages. As a result, instructions in previous dietary studies account only for contamination transfer from food packaging materials and preparation.
No studies have been found that succeed in quantitatively parsing the growing, production, and processing contamination from that of food contact materials and preparation.
Unlike clinical blood panels and mass spectrometer tests which have accepted standards, meals — even those using the same recipes — are rife with confounding factors to which no laboratory-quality standards have ever been addressed.
Few chefs, other than bakers, adhere to the precise requirements of recipes. And even bakers can be confounded by variations in measurements, sourced materials, timing, and baking temperatures.
This is a first effort at applying the basic principals of laboratory bench science protocols and practices to a kitchen responsible for dietary intervention meals.
Best bench science practices in the kitchen developed in a study of the effects of Bisphenol A and Inflammation.
Non-food exposures
Numerous non-food sources of BPA are present in the environment and likely pose significant “background” contamination which will vary depending upon the test subjects’ lifestyles and environments.
This is particularly critical because major health effects including estrogen/testosterone disruptions, inflammation, and metabolic events such as insulin resistance and lipid imbalance, can be affected by low doses of scores of chemicals.
For more on this see: Reducing Non-Food Exposures.
Sanitation/Hygiene
As with all appropriate bench science work, preparation surfaces should be non-reactive, non-porous, resistant to chemicals, heat, and easy to clean. Stone or ceramic tiles can accumulate contaminants in grout. Sealants will contain polymers. Polymer-based surfaces cannot be used.
Dietary interventions also require that preparation surfaces must be efficient to maintain hygiene in the event that food makes incidental contact.
Stainless steel offers all of those characteristics which is why professional and health-department approved kitchens are almost totally stainless steel.
Environment – Floor, walls, ventilation PM2.5
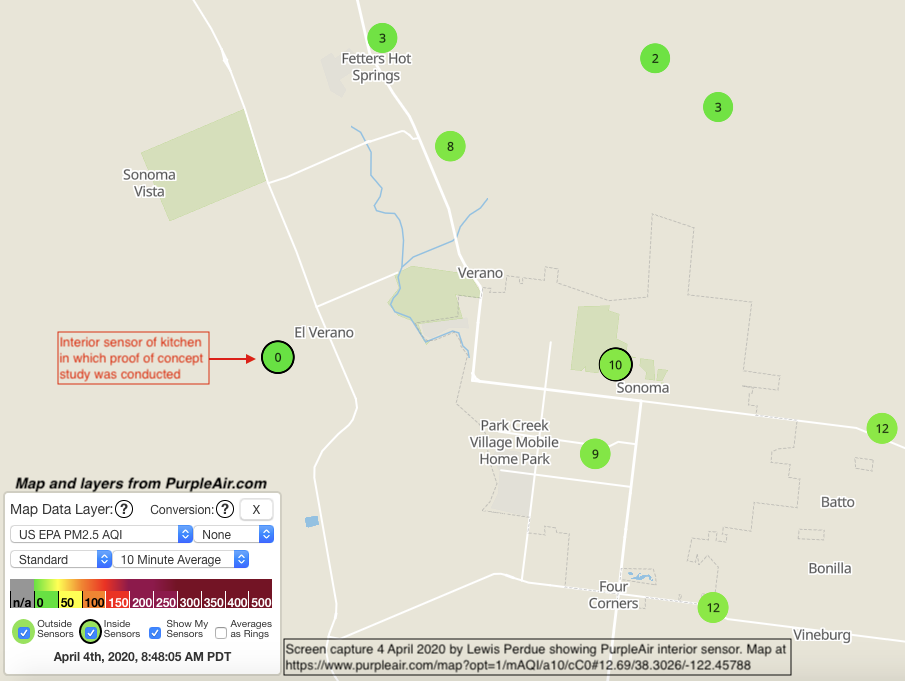
Constant ventilation of HEPA-filtered air is vital to prevent contamination of food with polymer-based particles which can confound results. Air should be constantly monitored throughout the trial and records kept.
The type and age of the HVAC system should be noted as well as the type of filter used and the ducting materials (metal versus plastic).
Immediately after dusting, the HVAC filter should be changed and the system fan run continuously for four hours to collect as much dust as possible which has been stirred up from cleaning. A fresh filter should then be installed.
HEPA-class air purifier use should report the make, model, capacity, and whether it is portable or installed with the HVAC system. If portable, the location should be noted along with the distance from the HVAC air return.
PurpleAir is an accurate and cost-effective solution for monitoring. Other than the cost of the sensor ($179), monitoring is free and historical data can be downloaded and saved.
This ad-free article is made possible by the financial support of the
Center for Research on Environmental Chemicals in Humans: a 501(c)(3) non-profit.
Please consider making a tax-deductible donation for continued biomedical research.
Cleaning Protocol – Floors
Polymer-based materials are universally found in professional and institutional kitchens given to their durability and ease of cleaning. However, friction against the floor caused by shoes and rolling carts inevitably creates ultra-fine particles many of which qualify as PM2.5 which — regardless of chemical composition — cause inflammation when inhaled. Significantly, many of the polymers from which flooring is made (such as polyvinyl chloride) have known health effects and can contaminate food destined for test subjects.
For this and standard hygiene reasons, floors should be wet-mopped with at the end of every food preparation day. The cleaner for this task and food preparation surfaces should be hypochlorite-based and free of surfactants such as ethoxylates that have been shown to cause health effects.
Spills and other unanticipated incidents should be cleaned with water — minus cleaning agents — when possible. Exceptions should be noted in the list of materials.
Cleaning Protocol – Preparation Surfaces
The same cleaning agents and protocols should be employed. No paper towels.
Towels – Sourcing and Washing
Only 100% cotton towels should be used. The source should be noted in the list of materials. Before first use, study towels should be washed and dried twice to remove any potential contaminants.
Towel, apron and other cloth washing
A new washer and dryer should be a purchased for a dietary intervention study because difficult-to-remove fabric softener and detergent residues can build up on the internal surfaces up from phthalates and other chemicals contained in detergents and fabric softeners. These can be basic, no-frills consumer-level, budget units. Note the brand/model of both.
Dishwashing
Replacing an institutional dishwasher is impractical and economically not necessary.
Before first use, dishwasher (note brand/model/age noted in equipment) should be run twice with an empty load using the detergent selected for the study. This will minimize deposits of undesirable chemicals.
The dishwasher should use the most vigorous setting (pots/pans), hottest final rinse. Do not use a heated dry cycle which may “bake on” organic contaminants. Instead, remove items immediately upon cycle finish when they are still wet. Then rinse all items under a running stream of carbon-filtered water and place in a stainless-steel dish rack to dry.
Age of clothes washers, dryers, and dishwashers is important
The age of clothes washers, dryers, and dishwashers with plastic components should be noted because polymer components — including seals — will leach more chemicals and additives as they age because many of the additives compounds are not covalently bound.
Equipment and Materials – Acceptable Materials
Plastic of any sort must be prohibited except in inevitable situations such as the seals in washers, dryers, and dishwashers. These utensils and items.
This includes so-called “BPA Free” items which are often manufactured with one or more of the 24 analogues of Bisphenol A and which have been found to exhibit similar harmful effects. This also includes the polymer Tritan which is a popular “BPA free” material that has also been shown to exhibit similar estrogenic, endocrine disrupting activity[vii].
Avoid any materials that are not glass or stainless steel. Exceptions may be made for the use of wooden cutting boards (with a minimum of glue joints) and utensils. Bamboo is not acceptable because the raw material is processed in an unknowable number of methods and the resins used are not disclosed.
Pots, pans, cooking preparation utensils, mixing bowls, measuring cups and spoons, eating utensils, serving plates, bowls, coffee/tea cups, beverage glasses should be either stainless steel or glass.
Cutting boards should be tight-grained maple composed of wide slats of wood to minimize glue joints are. Hand washing is required to avoid damage to the cutting board. Olive oil which is selected as a menu ingredient should be used to coat boards when dry. Do not use mineral oil or any other substance.
Note specific item name, model, and source in the equipment section.
Many basic glass items familiar to bench science (beakers, watch glasses, funnels, graduated cylinders) can facilitate meal preparations.
Soap and Detergents
Must avoid polymer ingredients as well as phthalates (including odor enhancers), surfactants with nonylphenol connections, nanoparticles, color enhancers, enzyme cleaners, fragrances.
Prohibited equipment and materials – Dishes, pots, pans, utensils, and items with any sort of plastic or non-stick coatings including ceramics
The following are forbidden:
- Sous vide
- non-stick pans
- most cooking oils
- plastic utensils
- plastic prep bowls
- synthetic gloves
- plastic bags
- plastic wrap
- Beverages in cans, plastic bottles, or glass.
Avoid food contact with plastics or recycled paper or cardboard. Recycled paper and cardboard contain Bisphenols because thermal cash register receipts are frequently included with other papers.
Avoid contact with any sort of box or wrapping with brightly colored graphics or type. Phthalates are contained in most packaging inks.
Do not wear any cloth garment or apron or use any towel or fabric with a design printed on them. Phthalates are contained in most of those design inks.
Food or other items that arrive in paper packaging should not be opened in the food preparation area.
Just as with plastic polymers, ceramic compositions are suspect.
No standards exist, nor any regulations for disclosure of constituents. Many contain chemical compounds that can leach out and contaminate foods, especially if they are predominantly alkaline or acidic. Some ceramic glazes can leach heavy metals such as lead, cadmium, and other colorant compounds.
So-called “stone” non-stick coatings have not been subjected to peer-reviewed investigations. However, these coatings are created by mixing ceramic or stone dust with a polymer. A slurry of those components is used to coat the interior of a cooking pot or pan. The moist coating is heated to a high temperature resulting in a non-stick surface.
Manufacturers are stubbornly non-transparent about materials because they consider the manufacturing process a trade secret.
Consumers and consumer groups have offered anecdotal opinions that cadmium or other heavy metals as well as organic polymer chemicals can leach into foods. Remember that the plural of anecdote is not data.
Very limited use should be made (and noted) for appliances such as a glass blender with a plastic seal at the bottom for steel blades. When used, aluminum foil should cover the mouth before inserting the plastic top.
Most aluminum foil used in cooking has a very light coating of release agent used in the rolling process. The identity and composition of that release agent is unknown <<cite + contact Reynolds>>
Food processors such as a Cuisinart cannot be used unless all components that contact food (including the bowl) are stainless steel or glass. We have been unable to locate a food processor with stainless steel bowl components. It is important to note that food processors marketed as “BPA” free — such as those like the Cuisinart with a Tritan bowl — leach estrogenic compounds that can alter blood profile measurements for inflammation and endocrine measurements.[vii]
Metal shredders, graters, or a mandoline are acceptable as long as all food contact surfaces are metal.
In addition, many food processor tasks (such as meat grinding) can be performed with a KitchenAid mixer which has a stainless steel bowl and attachments. Some KitchenAid attachments, such as the pasta maker, have plastic parts and should not be used. A manual, all-stainless pasta maker is preferred.
All cups, saucers, plates, bowls, serving, measuring, and storage containers should be glass. Measuring cups and spoons may be stainless steel
Also avoid:
- Electric drip coffee makers. Even those marketed as “BPA” free contain either BPA analogues that are jiust as harmbul or Tritan which has been found to be estrogenic like BPA. Electric and stove percolators, glass and steel French Press. Hand-poured drip using a glass carafe and either glass or metal grounds holder. A paper filter is not recommended because of unknown chemicals which can leach oit.
- Sodastream,
- Keurig and other “pod” beverage makers.
Plastic components of all those can leach chemicals of emerging concern.
Precise Measurements
Covered in this section: Best practices – Precise measurements.
Recipes are protocols
Cooking is a complicated organic chemical manufacturing process whose final product is determined by myriad processes that need to be controlled as tightly as possible and detailed notes taken at every step. New organic compounds are formed at every step.
Processing steps out of order, at an inappropriate temperature, prepared in the wrong manner, or cooked the wrong amount of time will affect the ultimate organic composition. In a non-study environment, that just affects the taste. But in a dietary intervention where very low levels of chemicals can affect physiological processes, the failure to follow the protocol may affect outcomes and replicability.
As one example, take spices: The process of “blooming” — heating spices in hot oil before adding other ingredients — will favor the extraction of lipid-soluble compounds that many cooks flavor for a particular intensity. Smaller particles offer different faces of the particles and greater surface area from which to extract the desired flavor compounds.
A precision recipe will approximate particle size (in millimeters or fractions of an inch). “Finely diced” is not precise. The amount of the spice, the temperature of the oil (in degrees, not “low heat”), and the elapsed time will alter — and determine — the chemical composition of the final product.
The next step in the protocol (recipe) will determine whether the oil and bloomed spices are to be reserved for later use (weight of final product noted) or whether other ingredients are to be added immediately after the elapsed blooming time. If meat is added at this point, one set of organic compounds will be created. If tomatoes, another variation; if potatoes yet another.
The variations are legion. If wine is added, the alcohol will extract a certain set of flavor compounds before evaporating. The remaining polyphenols and other compounds will react with others present to form another set of unknown compounds. Those compounds are a set of “unknowable unknowns.” Precision cooking protocols will help make those unknowable unknowns the same from trial to trial and increase the likelihood of replication if the protocols are followed exactly.
The important lesson is that each step and the timed order, temperature and method in which ingredients are added (“diced” is not precision. “half-inch cubes” (or “approximately a centimeter”) will all exert their own effects on the final organic manufacturing product called a meal.
Traditional American and English kitchen staff will be more familiar with non-metric quantities. Using English units for everything but weight may help avoid confusion among those also learning to cope with the demands of precision and documenting every step.
These steps were developed and tested in a small “proof-of-concept” dietary intervention to determine (among other things) that experienced cooks could adapt to the more precise demands with little trouble. That experience also showed that cooks with successful backgrounds in baking adapted most easily to the needed precision.
Ingredients as reagents
Each food element must be treated as a reagent. That means they must be exactly the same (or as close as possible) in:
- identity.
- purity,
- conformance with common food consumption practices
- consistency, availability, and capable of being reliably, economically, and easily obtained from sources accessible to other investigators.
Identity
Identity includes the vendor from which food is purchased. Preference should be granted for fruits, vegetables, grains, and other items that are branded and nationally available. Determining a varietal or cultivar identity and provenance transparency traceable to a specific and established source is desirable.
Obtaining the food directly from a nationally available grower/source who is willing to ship is the best option but may be seasonal. Farmers’ markets, while a valuable and healthy resource, are generally not acceptable unless their goods can be shipped nationally.
Purity
While the Organic Regulations set by the United States Department of Agriculture have been substantially degraded by the addition of scores of chemicals and pesticides, it is the only nationwide standard in the U.S.
Another consideration is the fact that most supermarket fruits and vegetables are irrigated with recycled municipal wastewater and are grown with commercial fertilizers. Furthermore, an increasing percentage are fertilized with biosolids — a euphemism for the human feces sludge from municipal sewage treatment plants once the recycled water has been removed.
Both biosolids and recycled wastewater are sources of undesirable unhealthy chemical and biological contaminants as well as micro- and nanoplastics, and their resulting plastic chemical contamination. (Micro- and Nanoplastics: Health Implications)
Currently, USDA organic regulations do not list biosolids and recycled wastewater in either its allowed or prohibited substances.
There have been efforts by large agricultural corporations to have those included as allowed. However, biosolids and recycled wastewater contain scores of prohibited substances including pharmaceuticals and illegal narcotics.
Conformance with common food consumption practices
For the broadest applicability, foods chosen for a dietary intervention study should reflect the most common consumption habits of the studied population. As one example, our proof-of-concept study attempted to emulate a “typical” American diet. To create that diet, investigators relied, upon the U.S. Department of Agriculture’s 2016 study: Americans’ Eating Patterns and Time Spent on Food.
We did not include most “niche” foods such as plant-based meat and milk substitutes because they are not mainstream dietary items. In addition, those are both highly processed items that experience substantial contact with plastic.
Consistency & Availability
Foods for a precision dietary capable of being reliably, economically, and easily obtained from sources accessible to other investigators.
While every effort should be made to reduce confounding factors — especially for chemicals targeted for examination in a study — extreme sourcing to avoid supply chain contamination can make a study more time-consuming, expensive, and non-replicable.
One example of an extreme sourcing (and ultimately impractical) protocol can be found at: Revised Stealth Syndromes Human Study Protocol – APPENDIX 2 – Detailed parameters of intervention diet selections. The effort to develop that selection protocol took more than six months — including personal visits to baseline food sources.
However, our proof-of-concept investigation demonstrated that the protocol’s extreme measures and emphasis on obtaining food directly from the source was impractical to implement fully. The time and expense, plus the seasonal variation in foods, would render such a study incapable of being replicated by other investigators.
Examples of Adjustments to Facilitate Replicability
The need for widespread availability for study foods demands national sourcing. That meant that — for our study — the food for the intervention leg of a study needed come from direct sources willing to ship food nationwide, or on large grocery chains.
To date, Whole Foods Markets are the only U.S. supermarket chain that bans its fruit and vegetable vendors from using biosolids.
Biosolids and recycled wastewater contain hundreds of chemical contaminants.Whole Foods has not specifically addressed the issue of recycled wastewater.
Despite that, we chose Whole Foods for most of the intervention study ingredients because of its biosolids ban and a wide variety of USDA-certified organic products.
We did test milk from Whole Foods against the Safeway brand and local organic brands. LC/MS spectrometry determined that four brands — Whole Foods, Organic Valley and two samples from a local dairy, Straus Family — all fell below the lab’s LOQ (o.03 micrograms/kilogram). We chose Organic Valley for milk and cheese because the brand is sold widely beyond Whole Foods.
Water
The quality of municipal water supplies varies greatly from place to place. Most contain measurable amounts of many chemicals of emerging concerns (such as Bisphenols, phthalates, and PFASs), as well as pharmaceuticals, and illegal drugs <<cite?>>
While research labs often have supplies of highly filtered water to remove chemical impurities, the kitchens of catering companies and institutional food facilities are usually filtered for taste and clarity only.
Unless a food preparation facility has a demand for multiple dietary studies that require expensive institutional filters, the most cost-effective approach is to install a residential-type, point-of-use filter.
Among many citations, a February 2020 study noted that: “All under-sink dual-stage and reverse osmosis filters tested showed near complete removal for all PFASs evaluated….In contrast, all other filters containing activated carbon exhibited variable PFAS removal. In these filters, PFAS removal efficiency was dependent on chain length.”
The most effective, according to the study, were the most expensive. Prices were not divulged in the study or supplemental material.
That study also did not assess the removal of bisphenol A or other chemicals. However, it did test a wide variety of filters, and including brand model names in the supplemental material. It is additionally significant because it explored the effects of molecular size on filtering efficiency. A further examination of molecular polarity — combined with size — may offer guidance on the ability to filter other substances.
An alternative selection method is to explore filters that have been NSF/ANSI certified for removal of specific chemicals involved in a study. As one example, our proof-of-concept study focused on BPA, and as a result, selected a Multi-Pure/Aquaversa under-sink, carbon-filter model which is NSF/ANSI 401 certified for “reduction of the compounds atenolol, bisphenol A, carbamazepine, DEET, estrone, ibuprofen, linuron, meprobamate, metolachlor, naproxen, nonylphenol, phenytoin, TECP, TCPP, and trimethoprim.”
The Aquaversa system for our proof-of-concept study cost approximately $650 installed.
Reagents as Ingredients
Sometimes laboratory reagents make for the best ingredients. Salt, baking powder, and baking soda are of unknown provenance. All contain undisclosed non-clumping chemicals and other substances of unknown identity, purity, and concentration.
We opted for laboratory reagents and decided to use baking powder instead of yeast in bread and similar recipes because of plastic packaging and because we could not determine what possible contaminants may have been introduced in production and packaging.
Our baking powder was compounded 2:1 mixture (by weight) of cream of tartar and baking soda.
<<photo>>
Supplemental Material
[i] Fasano, E., Bono-Blay, F., Cirillo, T., Montuori, P., and Lacorte, S. 2012. Migration of phthalates, alkylphenols, bisphenol A and di(2-ethylhexyl)adipate from food packaging. Food Control 27(1): 132-138.
[ii] Serrano, S.E., Braun, J., Trasande, L., Dills, R., and Sathyanarayana, S. 2014. Phthalates and diet: a review of the food monitoring and epidemiology data. Environmental Health 13: 43.
[iii] Guart, A., Bono-Blay, F., Borrell, A., and Lacorte, S. 2011. Migration of plasticizers phthalates, bisphenol A and alkylphenols from plastic containers and evaluation of risk. Food Additives & Contaminants Part A: Chemistry, Analysis, Control, Exposure & Risk Assessment 25(5): 676-685.
[iv] Bhunia, K., Sablani, S.S., Tang, J., and Rasco, B. 2013. Migration of chemical compounds from packaging polymers during microwave, conventional heat treatment, and storage. Comprehensive Reviews in Food Science and Food Safety 12(5): 523-545.
[v] Bang, D.Y., Kyung, M., Kim, M.J., Jung, B.Y., Cho, M.C., Choi, S.M., Kim, Y.W., Lim, S.K., Lim, D.S., Won, A.J., Kwack, S.J., Lee, Y., Kim, H.S., and Lee, B.M. 2012. Human risk assessment of endocrine-disrupting chemicals derived from plastic food containers. Comprehensive Reviews in Food Science and Food Safety 11(5): 453-470.
[vi] Groh, K.J., Geuke, B., and Muncke, J. 2017. Food contact materials and gut health: Implications for toxicity assessment and relevance of high molecular weight migrants. Food and Chemical Toxicology 109(1): 1-18.
[vii] Bittner, G.D., Denison, M.S., Yang, C.Z., Stoner, M.A., and He, G. 2014. Chemicals having estrogenic activity can be released from some bisphenol a-free hard and clear, thermoplastic resins. Environmental Health 13: 103.
[viii] Schecter, A., Lorber, M., Guo, Y., Wu, Q., Yun, S.H., Kannan, K., Hommel, M., Imran, N., Hynan, L.S., Cheng, D., Colacino, J.A., and Birnbaum, L.S. 2013. Phthalate concentrations and dietary exposure from food purchased in New York State. Environmental Health Perspectives 121(4): 473-479.
[ix] Cariou, R., Larvor, F., Monteau, F., Marchand, P., Bichon, E., Dervilly-Pinel, G., Antignac, J-P., and Le Bizec, B. 2016. Measurement of phthalates diesters in food using gas chromatography-tandem mass spectrometry. Food Chemistry 196: 211-219.
[x] Van Holderbeke, M., Geerts, L., Vanermen, G., Servaes, K., Sioen, I., De Henauw, S., and Fierens, T. 2014. Determination of contamination pathways of phthalates in food products sold on the Belgian market. Environmental Research 134: 345-352.
[xi] Fasano, E., Bono-Blay, F., Cirillo, T., Montuori, P., and Lacorte, S. 2012. Migration of phthalates, alkylphenols, bisphenol A and di(2-ethylhexyl)adipate from food packaging. Food Control 27(1): 132-138.
[xii] Cirillo, T., Latini, G., Castaldi, M.A., Dipaola, L., Fasano, E., Esposito, F., Scognamiglio, G., Di Francesco, F., and Cobellis, L. 2015. Exposure to di-2-ethyhexyl phthalate, di-N-butyl phthalate and bisphenol A through infant formulas. Journal of Agricultural and Food Chemistry 63(12): 3303-3310.
[xiii] Fierens, T., Vanermen, G., Van Holderbeke, M., De Henauw, S., and Sioen, I. 2012. Effect of cooking at home on the levels of eight phthalates in foods. Food and Chemical Toxicology 50(12): 4428-4435.
[xiv] Ionas, A.C., Dirtu, A.C., Anthonissen, T., Neels, H., and Covaci, A. 2014. Downsides of the recycling process: Harmful organic chemicals in children’s toys. Environment International 65: 54-62.
[xv] Hamrick, K., and McClelland, K. 2016. Americans’ Eating Patterns and Time Spent on Food: The 2014 Eating & Health Module Data. Economic Information Bulletin No. (EIB-158); 51pp.
[xvi] Guart, A., Wagner, M., Mezquida, A., Lacorte, S., Oehlmann, J., and Borrell, A. 2013. Migration of plasticizers from TritanTM and polycarbonate bottles and toxicological evaluation. Food Chemistry 141(1): 373-380.
[xvii] Bittner, G.D., Yang, C.Z., and Stoner, M.A. 2014. Estrogenic chemicals often leach from BPA-free plastic products that are replacements for BPA-containing polycarbonate products. Environmental Health 13: 41.
[xviii] Bittner, G.D., Denison, M.S., Yang, C.Z., Stoner, M.A., and He, G. 2014. Chemicals having estrogenic activity can be released from some bisphenol a-free hard and clear, thermoplastic resins. Environmental Health 13: 103.
[xix] Dolinoy, D.C., Huang, D., and Jirtle, R.L. 2007. Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. PNAS 104(32): 13056-13061.
[xx] Tarapore, P., Ying, J., Ouyang, B., Burke, B., Bracken, B., and Ho, S.M. 2014. Exposure to bisphenol A correlates with early-onset prostate cancer and promotes centrosome amplification and anchorage-independent growth in vitro. PLoS One 9(3): e90332.
[xxi] Pepys, M, B., and Hirschfield, G.M. 2003. C-reactive protein: A critical update. The Journal of Clinical Investigation 111(12): 1805-1812.
[xxii] Gao, H., Yang, B-J., Li, N., Feng, L-M., Shi, X-Y., Zhao, W-H., and Liu, S-J. 2015. Bisphenol A and Hormone-Associated Cancers: Current Progress and Perspectives. Medicine 94(1): e211.
[xxiii] Tarapore, P., Ying, J., Ouyang, B., Burke, B., Bracken, B., and Ho, S.M. 2014. Exposure to bisphenol A correlates with early-onset prostate cancer and promotes centrosome amplification and anchorage-independent growth in vitro. PLoS One 9(3): e90332.
[xxiv] Labbé, D.P., Zadra, G., Ebot, E.M., Mucci, L.A., Kantoff, P.W., Loda, M., and Brown, M. 2014. Role of diet in prostate cancer: the epigenetic link. Oncogene 34: 4683-4691.
[xxv] Fisher, M., Arbuckle, T.E., Mallick, R., LeBlanc, A., Hauser, R., Feeley, M., Koniecki, D., Ramsay, T., Provencher, G., Bérubé, R., and Walker, M. 2015. Bisphenol A and phthalate metabolite urinary concentrations: Daily and across pregnancy variability. Journal of Exposure Science & Environmental Epidemiology 25: 231-239.
[xxvi] Stahlhut, R.W., Welshons, W.V., and Swan, S.H. 2009. Bisphenol A data in NHANES suggests longer than expected half-life, substantial nonfood exposure, or both. Environmental Health Perspectives 117(5): 784-789.
[xxvii] Song, Y., Hauser, R., Hu, F.B., Franke, A.A., Liu, S., and Sun, Q. 2014. Urinary concentrations of bisphenol A and phthalate metabolites and weight change: a prospective investigation in US women. International Journal of Obesity 38(12): 1532-1537.
[xxviii] Fisher, M., Arbuckle, T.E., Mallick, R., LeBlanc, A., Hauser, R., Feeley, M., Koniecki, D., Ramsay, T., Provencher, G., Bérubé, R., and Walker, M. 2015. Bisphenol A and phthalate metabolite urinary concentrations: Daily and across pregnancy variability. Journal of Exposure Science & Environmental Epidemiology 25: 231-239.
[xxix] Larsson, K., Björklund, K.L., Palm, B., Wennberg, M., Kaj, L., Lindt, C.H., Jönsson, B.A.G., and Berglund, M. 2014. Exposure determinants of phthalates, parabens, bisphenol A and triclosan in Swedish mothers and their children. Environment International 73: 323-333.
[xxx] Hutter, K.P., Kundi, M., Hohenblum, P., Scharf, S., Shelton, J.F., Piegler, K., and Wallner, P. 2016. Life without plastic: A family experiment and biomonitoring study. Environmental Research 150: 639-644.
[xxxi] Ji, K., Lim Kho, Y., Park, Y., and Choi, K. 2010. Influence of a five-day vegetarian diet on urinary levels of antibiotics and phthalate metabolites: a pilot study with “Temple Stay” participants. Environmental Research 110(4): 375-382.
[xxxii] Galloway, T.S., Baglin, N., Lee, B.P., Kocur, A.L., Shepherd, M.H., Steele, A.M., BPA Schools Study Consortium, and Harries, L.W. 2018. An engaged research study to assess the effect of a “real-world” dietary intervention on urinary bisphenol A (BPA) levels in teenagers. BMJ Open 8: e018742.
[xxxiii] Rudel, R.A., Gray, J.M., Engel, C.L., Rawsthorne, T.W., Dodson, R.E., Ackerman, J.M., Rizzo, J., Nudelman, J.L., and Brody, J.G. 2011. Food packaging and Bisphenol A and Bis(2-Ethyhexyl) Phthalate exposure: Findings from a dietary intervention. Environmental Health Perspectives 119(7): 914-920.
[xxxiv] Sathyanarayana, S., Alcedo, G., Saelens, B.E., Zhou, C., Dills, R.L., Yu, J., and Lanphear, B. 2013. Unexpected results in a randomized dietary trial to reduce phthalate and bisphenol exposures. Journal of Exposure Science & Environmental Epidemiology 23: 378-384.
[xxxv] Sathyanarayana, S., Alcedo, G., Saelens, B.E., Zhou, C., Dills, R.L., Yu, J., and Lanphear, B. 2013. Unexpected results in a randomized dietary trial to reduce phthalate and bisphenol exposures. Journal of Exposure Science & Environmental Epidemiology 23: 378-384.
[xxxvi] Xue, J., Liu, W., and Kannan, K. 2017. Bisphenols, Benzophenones, and Bisphenol A Diglycidyl Ethers in Textiles and Infant Clothing. Environmental Science & Technology 51(9): 5279-5286.
[xxxvii] Sathyanarayana, S., Alcedo, G., Saelens, B.E., Zhou, C., Dills, R.L., Yu, J., and Lanphear, B. 2013. Unexpected results in a randomized dietary trial to reduce phthalate and bisphenol exposures. Journal of Exposure Science & Environmental Epidemiology 23: 378-384.
[xxxviii] Dodson, R.E., Nishioka, M., Standley, L.J., Perovich, L.J., Brody, J.G., and Rudel, R.A. 2012. Endocrine disruptors and asthma-associated chemicals in consumer products. Environmental Health Perspectives 120(7): 935-943.
[xxxix] Song, Y., Hauser, R., Hu, F.B., Franke, A.A., Liu, S., and Sun, Q. 2014. Urinary concentrations of bisphenol A and phthalate metabolites and weight change: a prospective investigation in US women. International Journal of Obesity 38(12): 1532-1537.
[xl] Bornehag, C-G., Lundgren, B., Weschler, C.J., Sigsgaard, T., Hagerhed-Engman, L, and Sundell, J. 2005. Pthalates in indoor dust and their association with building characteristics. Environmental Health Perspectives 113(10): 1399-1404.
[xli] Marshall, J. 2013. PM 2.5. PNAS 110(22): 8756.
[xlii] Schwartz, J., Laden, F., and Zanobetti, A. 2002. The concentration-response relation between PM(2.5) and daily deaths. Environmental Health Perspectives 110(10): 1025-1029.
[xliii] Pope III, C.A., Ezzati, M., Cannon, J.B., Allen, R.T., Jerrett, M., and Burnett, R.T. 2018. Mortality risk and PM2.5 air pollution in the USA: an analysis of a national prospective cohort. Air Quality, Atmosphere & Health 11(3): 245-252.
[xliv] Franklin, M., Zeka, A., and Schwartz, J. 2007. Association between PM2.5 and all-cause and specific-cause mortality in 27 US communities. Journal of Exposure Science & Environmental Epidemiology 17: 279-287.